Next-Generation DVT Thrombectomy
Retriever Medical is developing the ClotHound™ thrombectomy system for deep vein thrombosis (DVT).

Retriever Medical is developing the ClotHound™ thrombectomy system for deep vein thrombosis (DVT).

Deep vein thrombosis affects hundreds of thousands of patients annually.
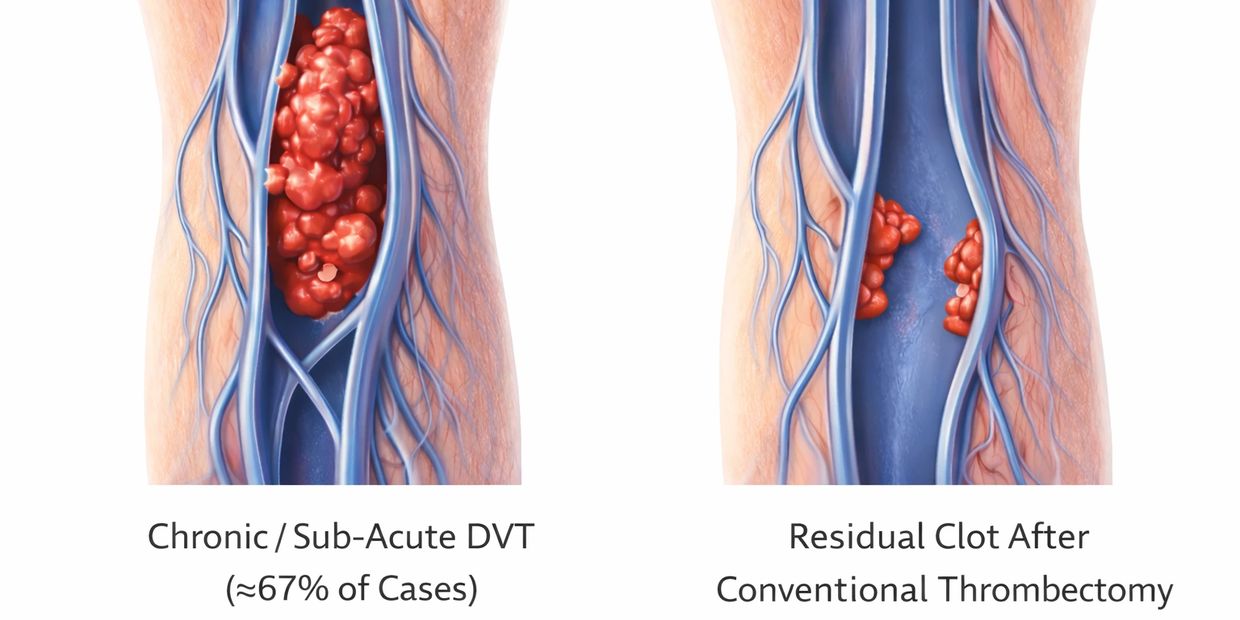
~67% of real-world cases involve sub-acute or chronic clot — not fresh thrombus.
Yet most thrombectomy systems are optimized for acute clot.
The result: residual clot, incomplete removal, and repeat procedures.

The ClotHound™ system deploys a distal sphere designed to anchor the device within the vessel while providing embolic protection during thrombectomy, helping reduce distal clot migration.

Once deployed, the device is engineered to mechanically engage acute, subacute, and chronic wall-adherent thrombus, allowing controlled clot capture within the treatment zone.

Following thrombus engagement, the captured clot is withdrawn through the catheter system, enabling efficient removal of thrombus from the venous system.
This animation illustrates the operating principles of the ClotHound™ thrombectomy system. The device is designed to mechanically engage acute, subacute, and chronic wall-adherent thrombus while supporting distal embolic protection during thrombectomy procedures.The system’s dual-sphere architecture enables controlled clot engagement and efficient thrombus removal from the venous system.
Independent preclinical testing demonstrated strong thrombus removal with distal embolic protection:
Results obtained from independent preclinical testing in a controlled porcine thrombectomy model.
The ClotHound™ system is being developed for 510(k) clearance for the treatment of deep vein thrombosis (DVT). Development is supported by independent preclinical validation and planned GLP animal studies designed to support regulatory submission.

Initial indication for the ClotHound™ system, targeting thrombus removal from the deep venous system of the lower extremities.

Future development may extend the platform to support thrombectomy procedures for pulmonary embolism within the pulmonary arteries.

The platform architecture may support treatment of additional venous conditions including iliocaval thrombosis and chronic venous occlusions.

We use cookies to analyze website traffic and optimize your website experience. By accepting our use of cookies, your data will be aggregated with all other user data.